Key Points
Diabetic Retinopathy = Diabetic microangiopathy affecting the retinal blood vessels leading to progressive retinal damage and ultimately blindness
- Occurs due to poor metabolic control
- Often the first complication to develop
- Can lead to complete blindness
Epidemiology & Aetiology
- Diabetic retinopathy is the commonest cause of blindness in <65 year olds
- Risk factors:
- Poor metabolic control chronically
- A small change in Hba1c has a massive impact
- Retinopathy affects both Type 1 and 2 diabetics, but is more noticeable T1DM
- Longer duration of diabetes
- After 20 years 90% type 1 and 60% type 2 will have diabetic retinopathy
- Hypertension
- Hyperlipidaemia
- Anaemia
- Smoking
- Pregnancy
- Poor metabolic control chronically
Pathophysiology
- Damage in the eye is classed in two types:
- Retinopathy – vessel damage outside of the macula
- Maculopathy – vessel damage affecting the macula
- The implications of both are different
- Retinopathy develops through two main mechanisms
- Occlusion of the microcirculation leads to retinal ischaemia which results in:
- Microaneurysms – these rupture/leak to cause haemorrhages, exudates, etc
- Infarction of retinal tissue
- This is seen as Cotton Wool Spots which indicate axonal debris
- Neovascularisation and AV shunting occurs due to VEGF release
- Leakage from the microcirculation develops following high blood flow and occlusion. This causes:
- Haemorrhages due to blood leak
- Dot haemorrhages appear first
- BlotHaemorrhages- larger haemorrhages
- Flame-shaped haemorrhages
- Oedema due to fluid leak
- Exudates due to lipid/lipoprotein leak
- Haemorrhages due to blood leak
- Occlusion of the microcirculation leads to retinal ischaemia which results in:
- This figure (Zaki et al. 2016) summarises the above process
Maculopathy: pathophysiology
Maculopathy is essentially retinopathy affecting the capillaries around the area of the macula/fovea
- This is the main cause of blindness in retinopathy
- There are 3 types
- Features are similar
Clinical Features
- Symptoms – these are usually absent until maculopathy or R3 (i.e. advanced disease)
- Blurred vision develops with maculopathy
- i.e. loss of visual acuity
- Floaters with vitreous humour changes in consistency
- Sudden vision loss with retinal detachment
- Blindness with maculopathy/vitreous haemorrhages/retinal detachment
- Blurred vision develops with maculopathy
- Other features may develop :
- Eye pain
- Reddening of the iris
- Irregular pupil
- Signs – See grading
Grading
Complications
- Non-retinopathic
- Cataracts – opacification of the lens with several mechanisms
- Juvenile ‘Snowflake’ cataracts are seen in diabetes due to osmotic changes in the lens from acute hyperglycaemia
- Age-related cataracts occur earlier and progress faster in diabetics
- May also develop retinal detachment cataracts
- May be reversible if hyperglycaemia is reduced
- POA Glaucoma is also more common due to diabetes
- Cataracts – opacification of the lens with several mechanisms
- Retinopathic- these occur after R3:
- Retinal vein occlusion – leads to oedema and swelling of the optic disc
- Rubeosis iris – neovascularisation of the iris due to anterior segment ischaemia
- Can lead to rubeotic glaucoma as the vessels can fibrose to block off the angle
- NB non-retinopathic glaucoma is also more common in diabetes
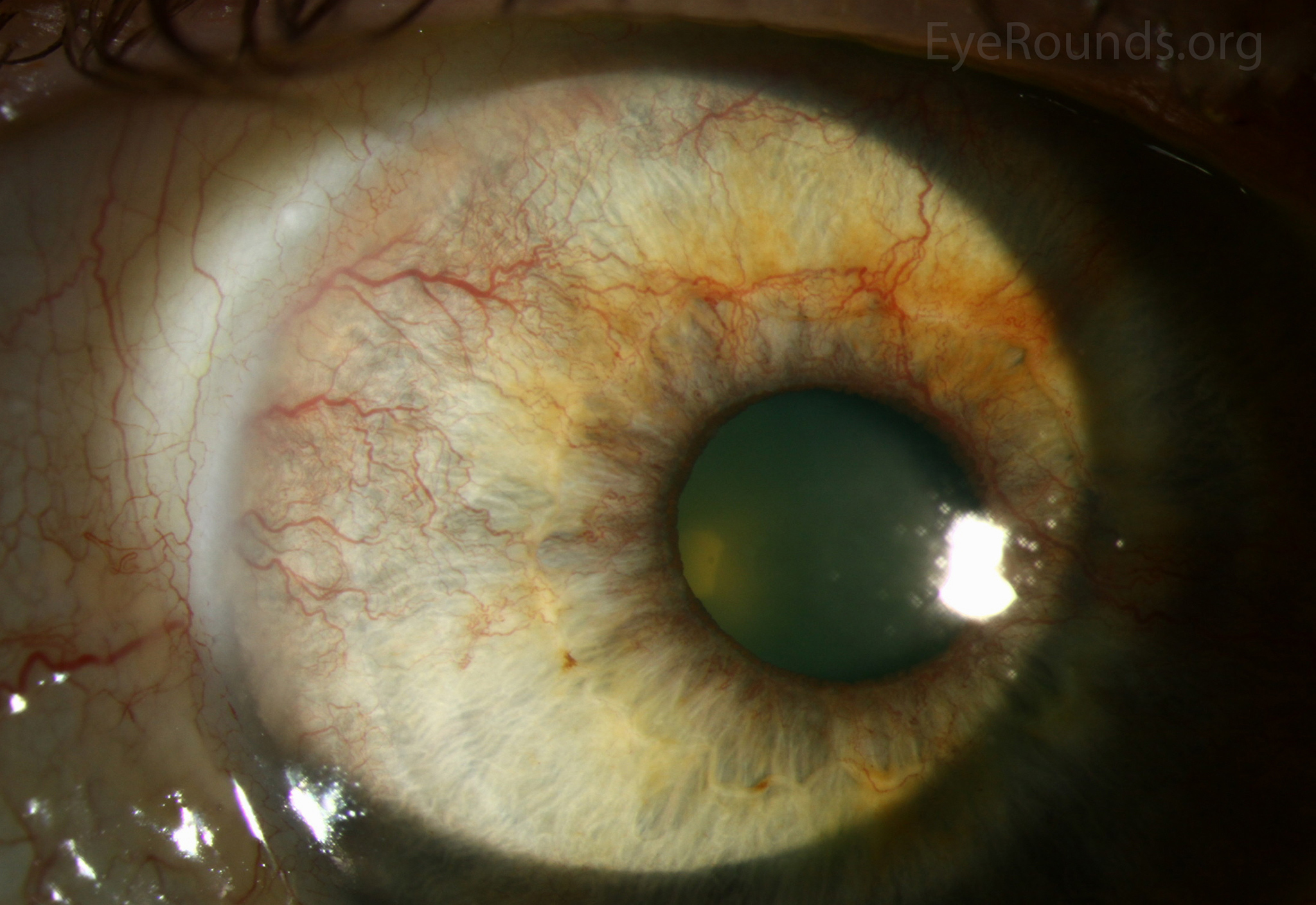 Image is from the Department of Ophthalmology and Visual Sciences, University of Iowa
Image is from the Department of Ophthalmology and Visual Sciences, University of Iowa
- Preretinal haemorrhages – bleed in the posterior hyaloid of the vitreous in front of the retina
- Show a fluid level because they are contained
- Vitreous haemorrhage – bleed within the vitreous humour due to permeable new blood vessels within the vitreous humour which can then bleed
- Can cause blindness
- Look like a blood haze in front of the retina
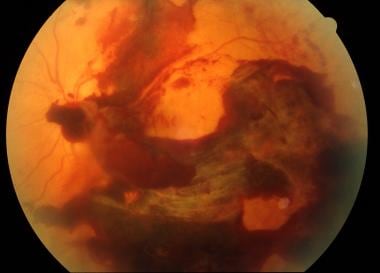 (image from Medscape eMedicine )
(image from Medscape eMedicine )
- Retinal detachment– fibrosis can cause tractiondetachment
- Blindnessoccurs if the maculais involved
- This is the latest to develop of all complications
- Must be treated with a vitrectomy
- Nerve palsies due to ischaemia
- Optic neuropathy occurs due to ischaemia of CNII
- CNIII palsy
- Blindness occurs mainly due to macula involvement but also inretinal detachmentand vitreous haemorrhages
Investigations
There are several things to look at :
- Assess visual acuity
- Shows maculopathy
- E.g. using a Snellen chart
- Correct for best corrective vision therefore can differentiate between refractive error and pathology. I.e use glasses or pinhole
- Dilated eye exams
- Opthalmoscopy – less often used now
- Retinal photography – required for screening
- 2 photographs are taken
- Disc centred
- Macula centred
- Both are analysed and graded by the technician
- 2 photographs are taken
- Slit lamp biomicroscopy – used to assess patients as it can penetrate through cataracts unlike photography
- OCT scanning
- Shows topography of the retina and thus allows abnormalities to be picked up
- 7 layers of the retina
- Often used for the macula but can be used for the rest of the retina
- Used in patients who have maculopathy on photography
- Shows oedema well
- Can identify other pathological features:
- E.g. dot and blot haemorrhages occur in the middle layers
- Fluorescein angiography
- Finds leakages in detail
- Allows identification of neovascularisation
- Useful for planning laser photocoagulation
Management
Prevention
- Aim to control diabetes and risk factors to prevent onset and progression
- Hba1c control
- BP control – typically using an ACEI
- Cholesterol control – statin for 1º prevention
- Correct anaemia
- Ensure all diabetic patients are referred to retinal screening
- Annual ophthalmoscopy
Management
- See referral stages above (diabetic team => HES referral => urgent HES referral)
Conservative
- Optimise glycaemic control
- Eye aids could help with visual impairment patients
Medical
- Fenofibrate is also beginning to be used
- Was mainly an anticholesterol drug but there is evidence for preventative benefit of retinopathy
- Intravitreal Injections
- VEGF inhibitors (e.g. Leucentis, aflibercept, bevacizumab)
- Reduce neovascularisation
- Used in diabetic maculopathy but also central retinal vein occlusion and ARMD
- Steroid injections
- Can be used to reduce macular oedema
- Mainly used in patients who have already had cataract replacements and are not at risk of glaucoma
- VEGF inhibitors (e.g. Leucentis, aflibercept, bevacizumab)
Surgical
- Retinal photocoagulation
- Laser peripheral retina to reduce the ischaemic area and thus reduce the release of vasoproliferative growth factors that induce neovascularisation
- Used in R3/M1
- 3 strategies are used
- PRP is the main one for R3
- The other two strategies are used for Maculopathy
- Focal for smaller areas – focal maculopathy
- Grid treatment is used for diffuse maculopathy
- However it can relapse therefore follow-up is required
- Vitrectomy
- Used to clean out vitreal bleeds and replace vitreous gel with silicone to prevent neovascularisation
